Description
| McKesson # | 504615 |
| Manufacturer # | SMM-5536 |
| Brand | Maxon™ |
| Manufacturer | Covidien |
| Application | Suture with Needle |
| Coating | Uncoated |
| Material Type | Monofilament Polyglyconate |
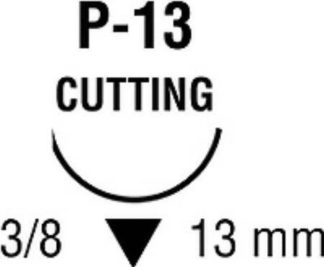
| Needle 1 Code | P-13 |
| Needle Length | 13 mm Length |
| Needle Material | Alloy Needle |
| Needle Shape | 3/8 Circle |
| Needle Type | Reverse Cutting Needle |
| Number of Needles | 1-Needle |
| Size | Size 4 – 0 |
| Sterility | Sterile |
| Suture Color | Clear Suture |
| Suture Length | 18 Inch Suture |
| Suture Type | Absorbable Uncoated |
| UNSPSC Code | 42312201 |
Features
- MAXON™ and MAXON™ CV synthetic absorbable sutures are prepared from polyglyconate, a copolymer of glycolic acid and trimethylene carbonate
- MAXON™ and MAXON™ CV synthetic absorbable sutures are indicated for use in general soft tissue approximation and/or ligation, including use in pediatric cardiovascular tissue, where growth is expected to occur, and in peripheral vascular surgery
- MAXON™ and MAXON™ CV synthetic absorbable sutures are not indicated for use in adult cardiovascular tissue, ophthalmic surgery, microsurgery and neural tissues
- MAXON™ sutures retain approximately 75% of initial tensile strength at two weeks, 65% of at three weeks and 50% at four weeks post-implant
- Absorption of MAXON™ sutures is minimal until about 60 days after implant and is essentially complete within six months
- This product is required to be reported under California Proposition 65